Understand the critical synergy between pH levels and residual chlorine effectiveness. Learn how to stay compliant with 2026 water quality regulations using precision monitoring solutions.
As we move through 2026, global water quality regulations have become more stringent than ever. One common pitfall in water disinfection is monitoring residual chlorine in isolation. To ensure true safety and compliance, understanding the relationship between pH and chlorine is essential. For many facilities, implementing an ERUN-SZ3-12 is the only way to manage this synergy automatically and stay ahead of regulatory requirements.
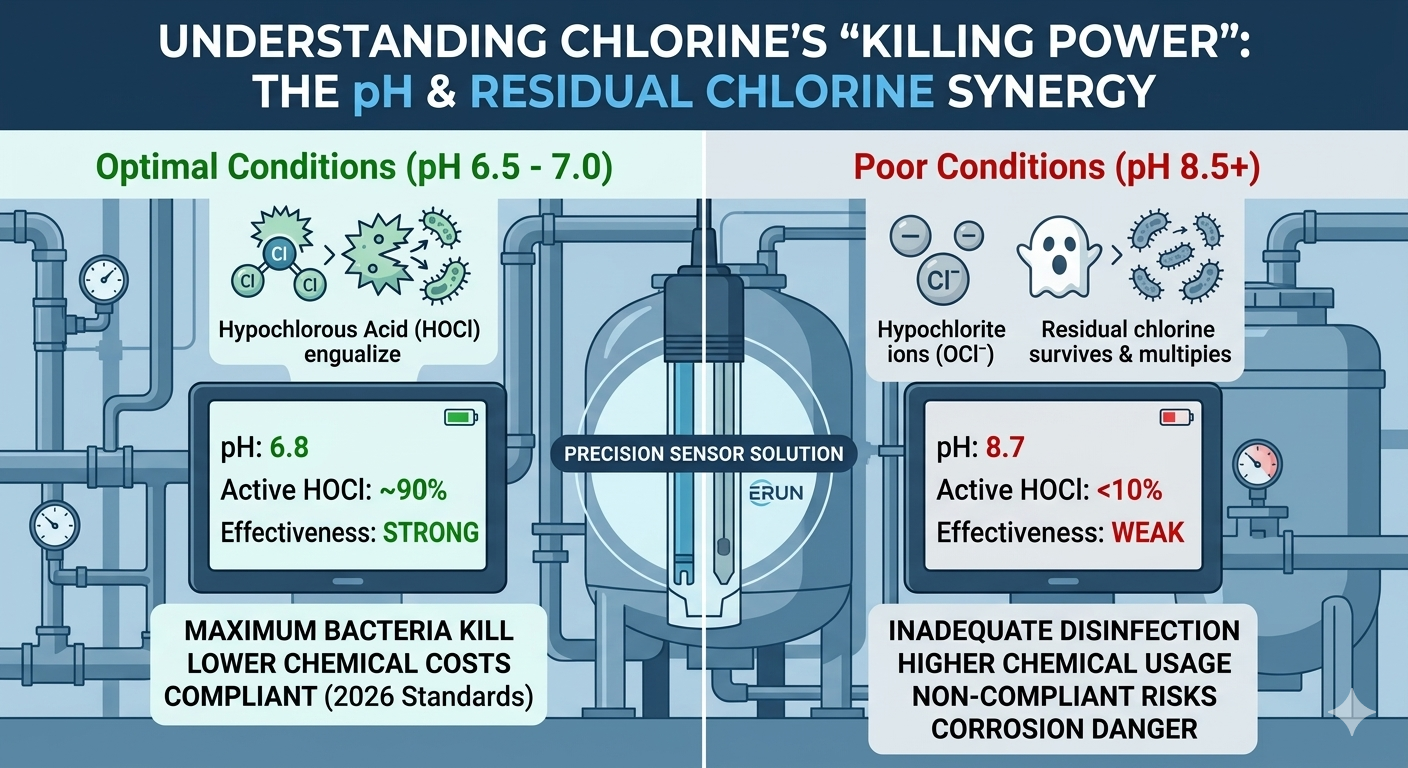
Chlorine disinfection effectiveness is highly pH-dependent. When chlorine is added to water, it forms Hypochlorous Acid (HOCl)—the active disinfecting agent.
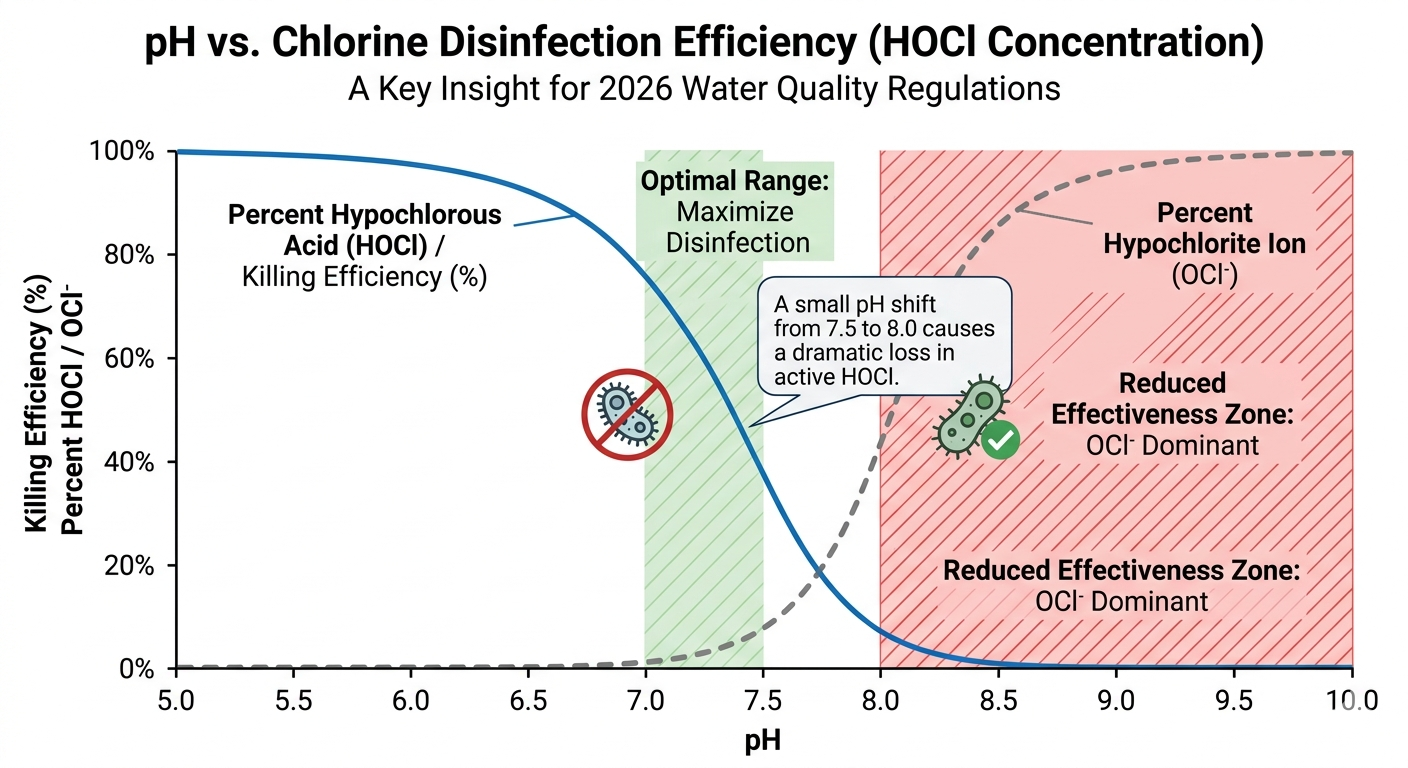
At pH 6.5-7.0: Most of the chlorine remains as highly effective HOCl.
At pH 8.5+: The chlorine shifts to Hypochlorite ions (OCl⁻), which are up to 80 times less effective at killing bacteria.
Without precise pH monitoring, your residual chlorine readings may be high, but your water could still be biologically unsafe.
Regulatory bodies now demand more than just "sufficient chlorine." They require proof of "effective disinfection."
Preventing Infrastructure Damage: Proper pH balance prevents the corrosive effects of overly acidic water on municipal pipes.
Optimizing Chemical Costs: By maintaining the ideal pH range, facilities can reduce the amount of chlorine required, lowering both costs and the formation of harmful disinfection byproducts (DBPs).

Modern B2B industrial engineering demands high-stability electrodes that can withstand harsh environments. Utilizing an integrated analyzer that provides temperature-compensated pH and chlorine data ensures that your facility remains compliant and your water remains crystal clear.
In 2026, precision is the only path to compliance. By integrating advanced sensors that account for the pH-chlorine synergy, industrial and municipal operators can guarantee water safety with absolute confidence.